Difference between revisions of "Electrolyte"
From Nordan Symposia
Jump to navigationJump to search (Created page with 'File:lighterstill.jpgright|frame *[http://en.wikipedia.org/wiki/19th_century 1834] ==Definitions== *1: a nonmetallic electric conductor in which curr...') |
m (Text replacement - "http://" to "https://") |
||
| Line 1: | Line 1: | ||
[[File:lighterstill.jpg]][[File:Lytes.jpg|right|frame]] | [[File:lighterstill.jpg]][[File:Lytes.jpg|right|frame]] | ||
| − | *[ | + | *[https://en.wikipedia.org/wiki/19th_century 1834] |
==Definitions== | ==Definitions== | ||
| − | *1: a nonmetallic [[electric]] conductor in which current is carried by the movement of [ | + | *1: a nonmetallic [[electric]] conductor in which current is carried by the movement of [https://en.wikipedia.org/wiki/Ions ions] |
2a : a substance that when [[dissolved]] in a suitable solvent or when fused becomes an ionic conductor | 2a : a substance that when [[dissolved]] in a suitable solvent or when fused becomes an ionic conductor | ||
| − | :b : any of the ions (as of [ | + | :b : any of the ions (as of [https://en.wikipedia.org/wiki/Sodium sodium] or [https://en.wikipedia.org/wiki/Calcium calcium]) that in biological fluid regulate or affect most metabolic processes (as the flow of nutrients into and waste products out of [[cells]]) |
==Description== | ==Description== | ||
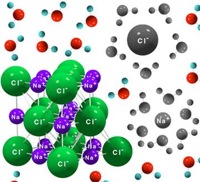
| − | Substance that conducts [[electric]] current as a result of dissociation of its [[molecules]] into positively and negatively charged particles called ions. The most familiar electrolytes are acids, bases, and salts, which ionize when dissolved in polar solvents such as [[water]]. Many salts, including sodium chloride, behave as electrolytes when melted in the [[absence]] of solvent, since they have ionic bonds. The most commonly used electrolytes are dissolved metal salts (for electroplating metals) and acids (in electric batteries). See also [ | + | Substance that conducts [[electric]] current as a result of dissociation of its [[molecules]] into positively and negatively charged particles called ions. The most familiar electrolytes are acids, bases, and salts, which ionize when dissolved in polar solvents such as [[water]]. Many salts, including sodium chloride, behave as electrolytes when melted in the [[absence]] of solvent, since they have ionic bonds. The most commonly used electrolytes are dissolved metal salts (for electroplating metals) and acids (in electric batteries). See also [https://en.wikipedia.org/wiki/Electrolysis electrolysis]. |
[[Category: Chemistry]] | [[Category: Chemistry]] | ||
Latest revision as of 00:43, 13 December 2020
Definitions
2a : a substance that when dissolved in a suitable solvent or when fused becomes an ionic conductor
- b : any of the ions (as of sodium or calcium) that in biological fluid regulate or affect most metabolic processes (as the flow of nutrients into and waste products out of cells)
Description
Substance that conducts electric current as a result of dissociation of its molecules into positively and negatively charged particles called ions. The most familiar electrolytes are acids, bases, and salts, which ionize when dissolved in polar solvents such as water. Many salts, including sodium chloride, behave as electrolytes when melted in the absence of solvent, since they have ionic bonds. The most commonly used electrolytes are dissolved metal salts (for electroplating metals) and acids (in electric batteries). See also electrolysis.